Synthesis of fluorinated amino acid derivatives through late-stage deoxyfluorinations - ScienceDirect

Synthesis of fluorinated amino acid derivatives through late-stage deoxyfluorinations - ScienceDirect

Practical Synthesis of Ethyl 3-Fluoro-1-pyrrole-2-carboxylate: A Key Fragment of a Potent Drug Candidate against Hepatitis B Virus | Organic Process Research & Development

Synthesis of Fluorine‐Containing Molecular Entities Through Fluoride Ring Opening of Oxiranes and Aziridines - Remete - 2019 - European Journal of Organic Chemistry - Wiley Online Library

Latest Highlights in Liquid-Phase Reactions for Organic Synthesis in Microreactors | Organic Process Research & Development

Synthesis of fluorinated amino acid derivatives through late-stage deoxyfluorinations - ScienceDirect

5-Aminopyrazole as precursor in design and synthesis of fused pyrazoloazines. - Abstract - Europe PMC

Synthesis of Fluorine‐Containing Molecular Entities Through Fluoride Ring Opening of Oxiranes and Aziridines - Remete - 2019 - European Journal of Organic Chemistry - Wiley Online Library
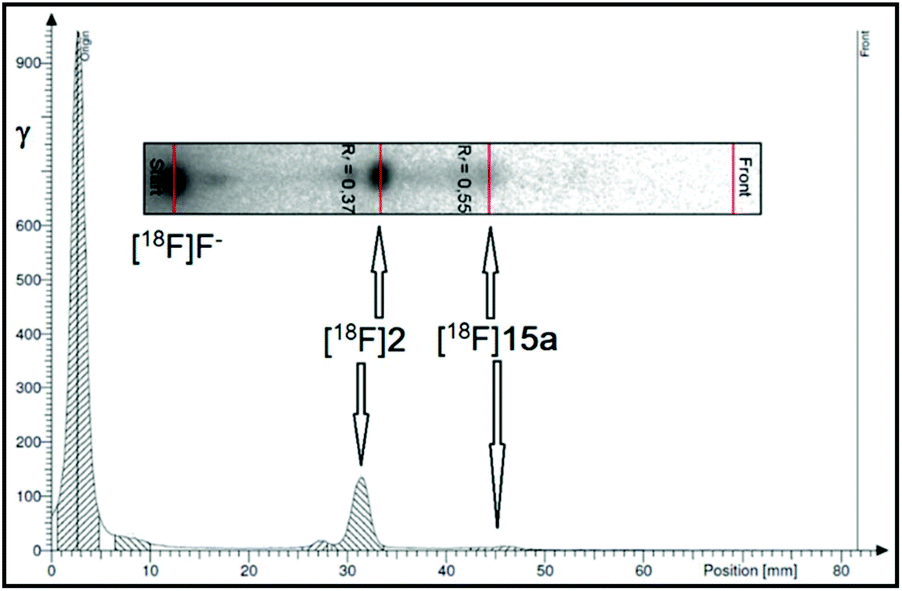
Synthesis, radiolabelling and initial biological characterisation of 18 F-labelled xanthine derivatives for PET imaging of Eph receptors - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00391C

Synthesis of fluorinated amino acid derivatives through late-stage deoxyfluorinations - ScienceDirect

Emulating Natural Product Conformation by Cooperative, Non‐Covalent Fluorine Interactions - Scheidt - 2017 - Chemistry – A European Journal - Wiley Online Library

5-Aminopyrazole as precursor in design and synthesis of fused pyrazoloazines. - Abstract - Europe PMC

Emulating Natural Product Conformation by Cooperative, Non‐Covalent Fluorine Interactions - Scheidt - 2017 - Chemistry – A European Journal - Wiley Online Library
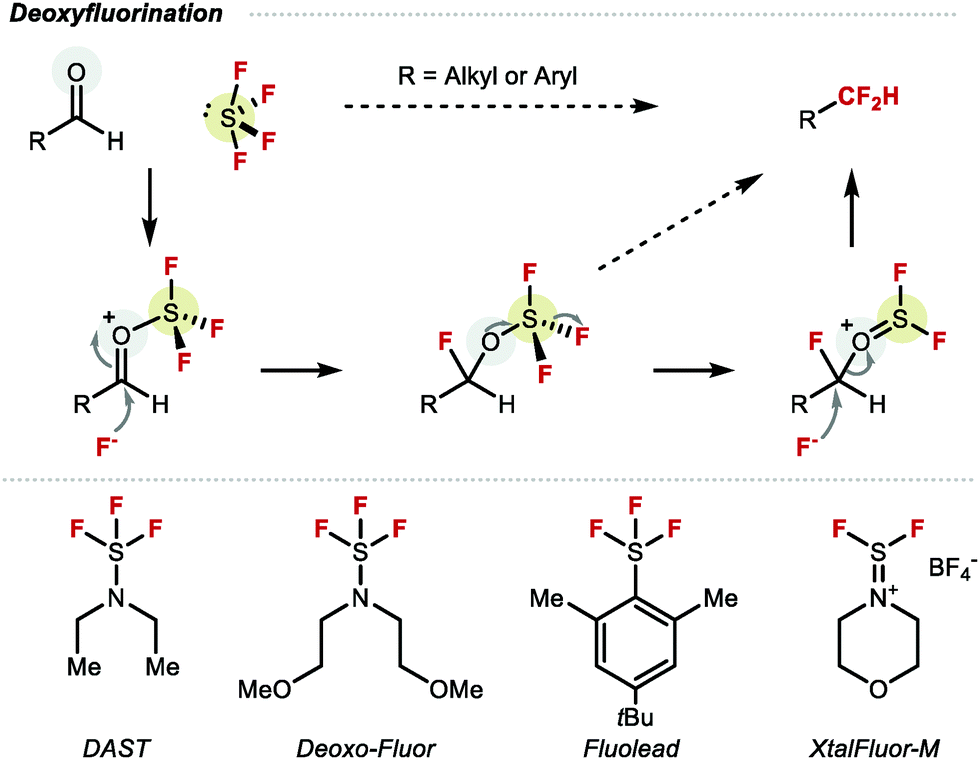
Late-stage difluoromethylation: concepts, developments and perspective - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D1CS00360G

Morpholinosulfur Trifluoride (Morph‐DAST)‐Mediated Rearrangement in the Fluorination of Cyclic α,α‐Dialkoxy Ketones toward 1,2‐Dialkoxy‐1,2‐difluorinated Compounds - Surmont - 2010 - Advanced Synthesis & Catalysis - Wiley Online Library