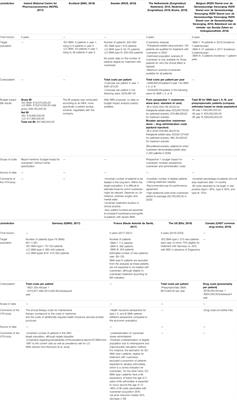
Frontiers | Assessing the Value of Nusinersen for Spinal Muscular Atrophy: A Comparative Analysis of Reimbursement Submission and Appraisal in European Countries | Pharmacology
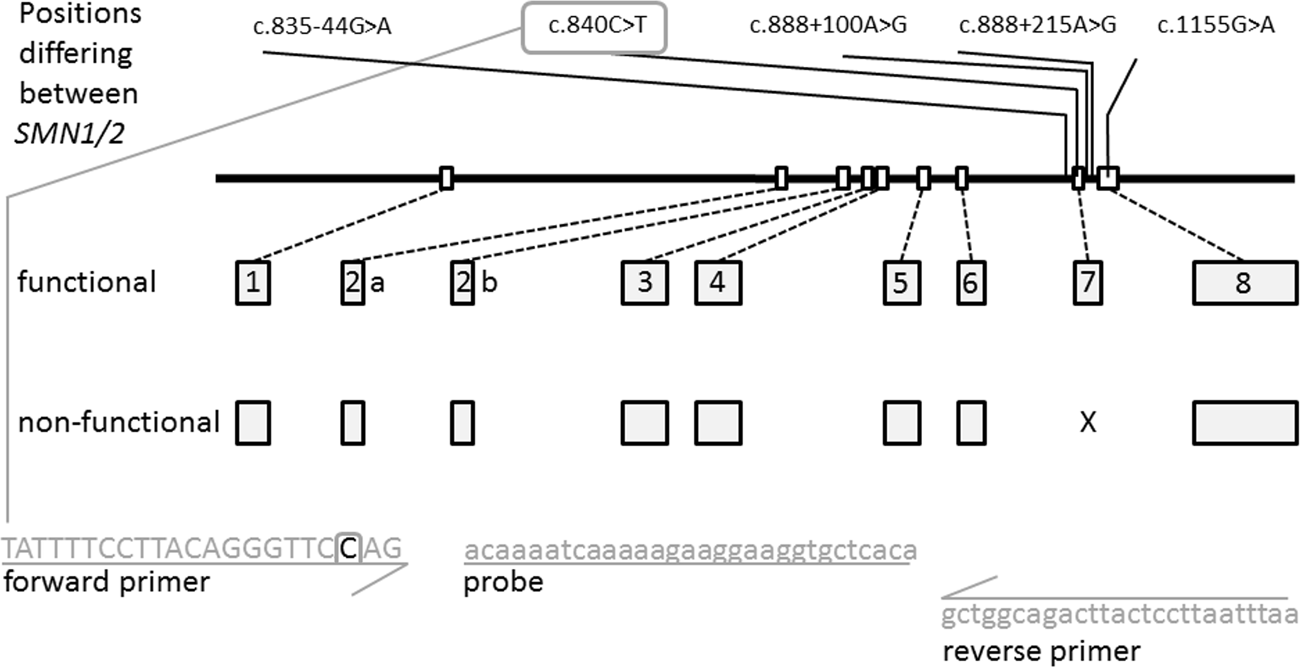
High-throughput genetic newborn screening for spinal muscular atrophy by rapid nucleic acid extraction from dried blood spots and 384-well qPCR | European Journal of Human Genetics
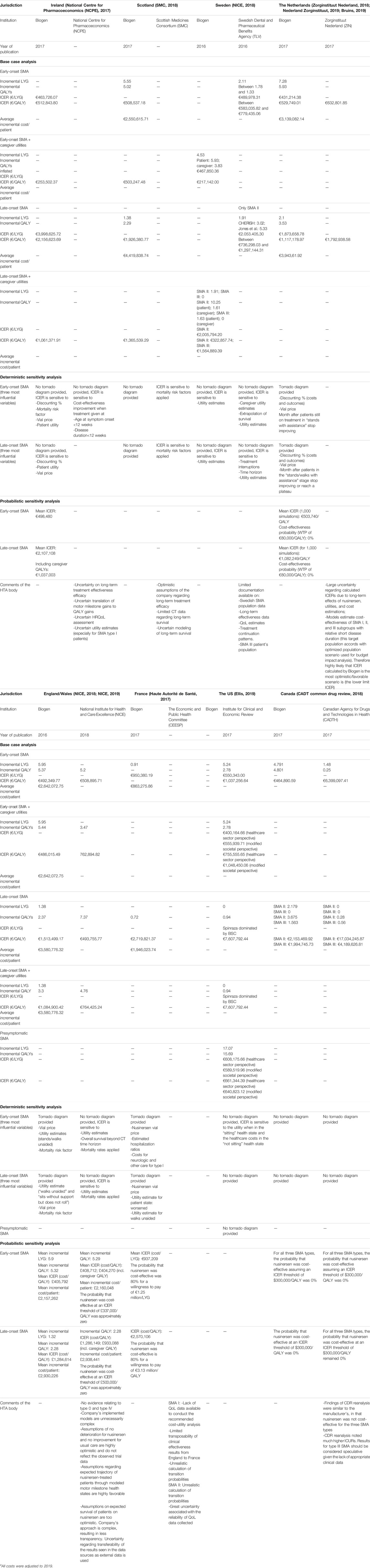
Frontiers | Assessing the Value of Nusinersen for Spinal Muscular Atrophy: A Comparative Analysis of Reimbursement Submission and Appraisal in European Countries | Pharmacology

DUX4 Transcript Knockdown with Antisense 2′-O-Methoxyethyl Gapmers for the Treatment of Facioscapulohumeral Muscular Dystrophy - ScienceDirect
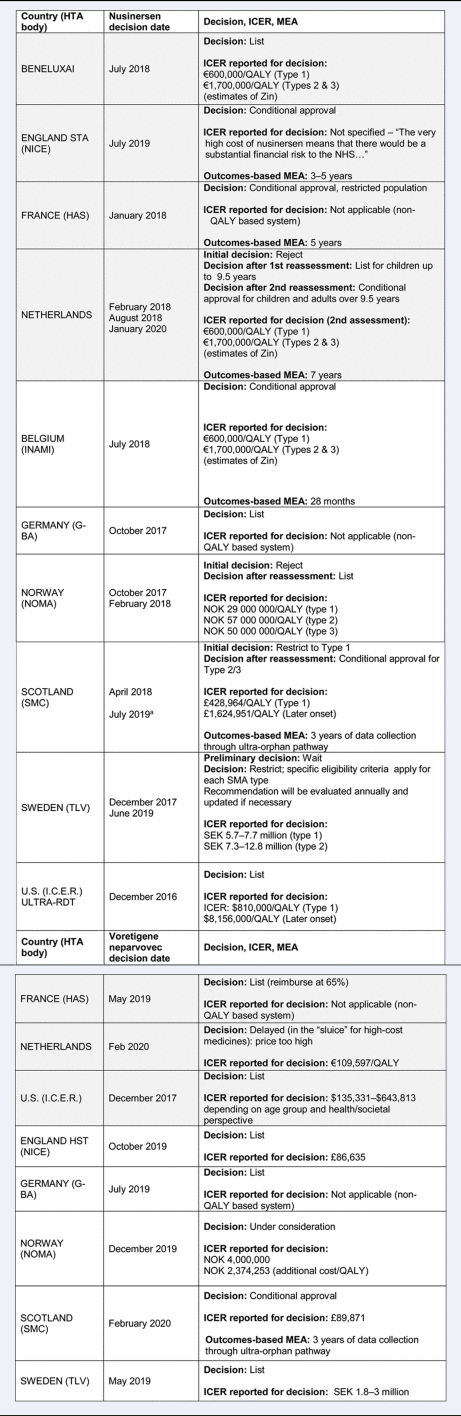
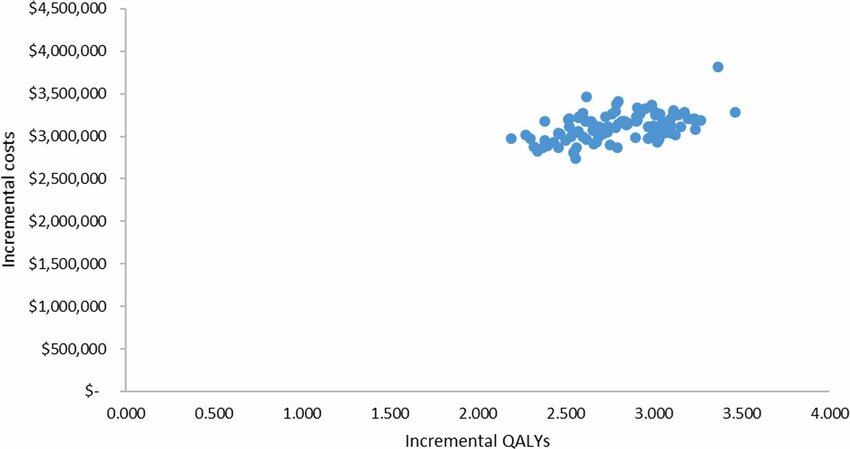
Examining the impact of different country processes for appraising rare disease treatments: a case study analysis | International Journal of Technology Assessment in Health Care | Cambridge Core

Oligonucleotide therapeutics: How to measure plasma concentration, tissue distribution and immunogenicity?

Constantin d'Ydewalle - Senior Scientist, Neuroscience Drug Discovery - The Janssen Pharmaceutical Companies of Johnson & Johnson | LinkedIn
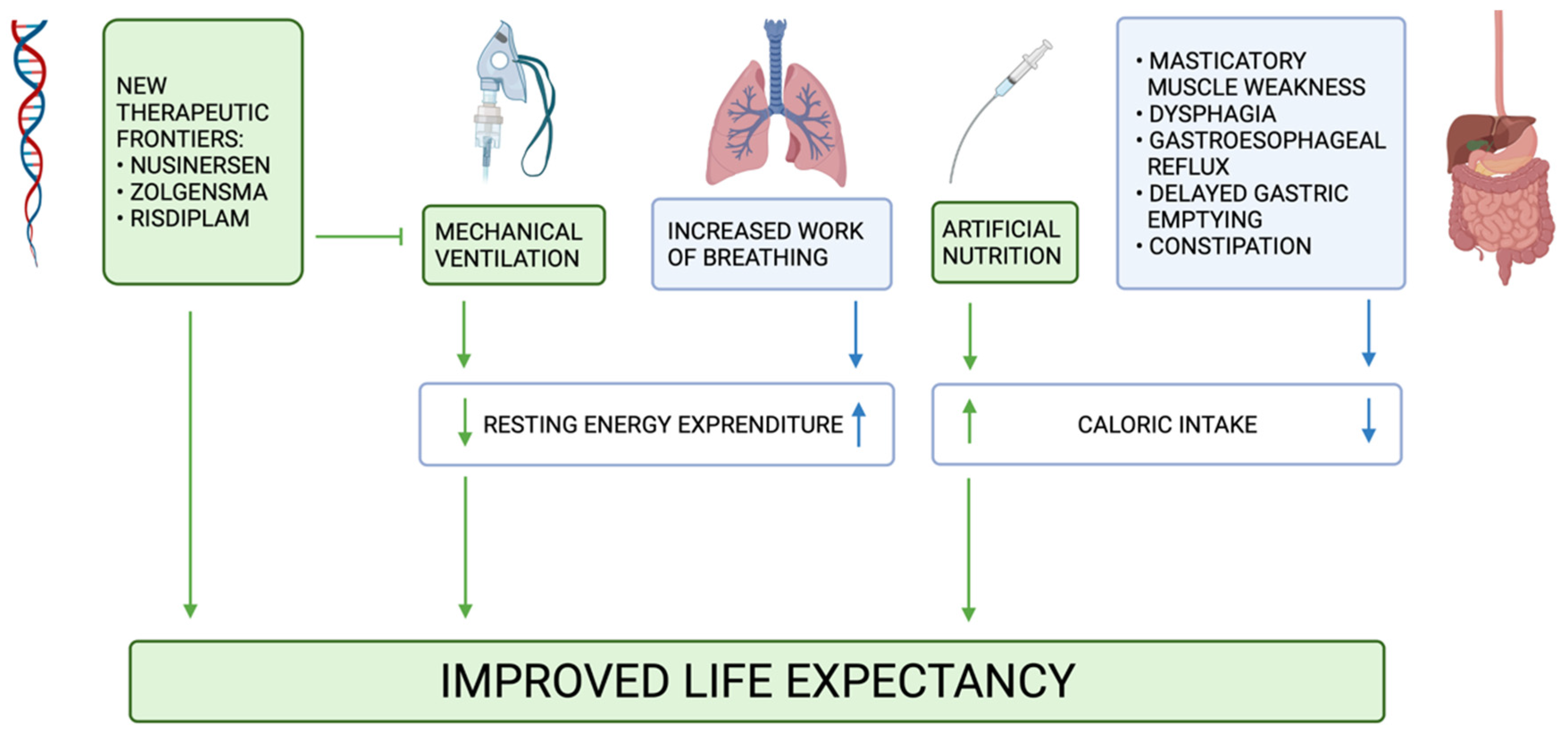
Nutrients | Free Full-Text | Nutritional, Gastrointestinal and Endo-Metabolic Challenges in the Management of Children with Spinal Muscular Atrophy Type 1 | HTML

Nusinersen expanded access program (EAP) enrollment. The nusinersen EAP... | Download Scientific Diagram

Nusinersen expanded access program (EAP) enrollment. The nusinersen EAP... | Download Scientific Diagram
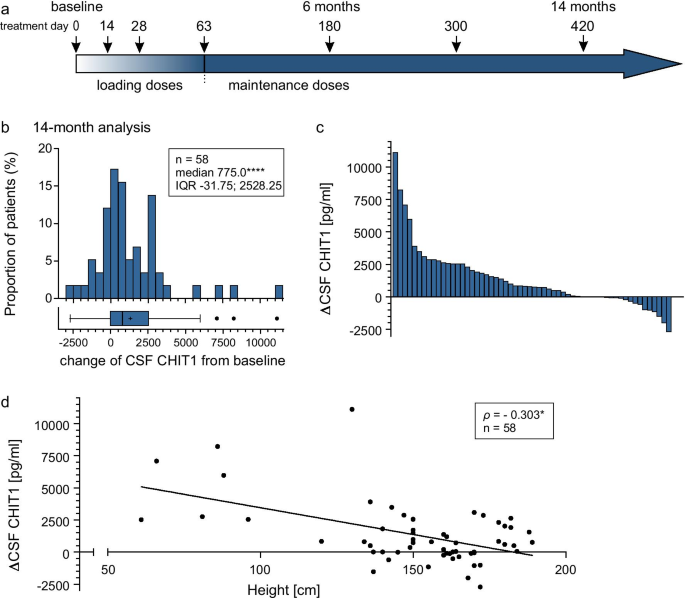
Increased chitotriosidase 1 concentration following nusinersen treatment in spinal muscular atrophy | Orphanet Journal of Rare Diseases | Full Text
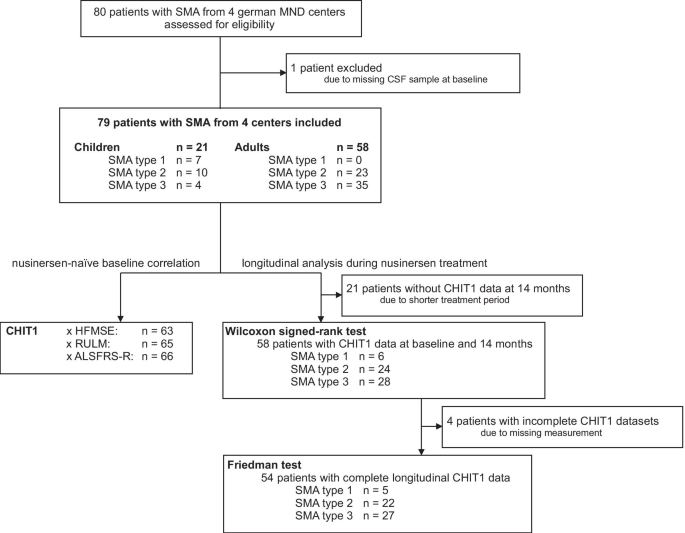
Increased chitotriosidase 1 concentration following nusinersen treatment in spinal muscular atrophy | Orphanet Journal of Rare Diseases | Full Text

Current evidence for treatment with nusinersen for spinal muscular atrophy: a systematic review | SpringerLink